The project was split up into 12 work packages, read more about what these involved below.
Content Table
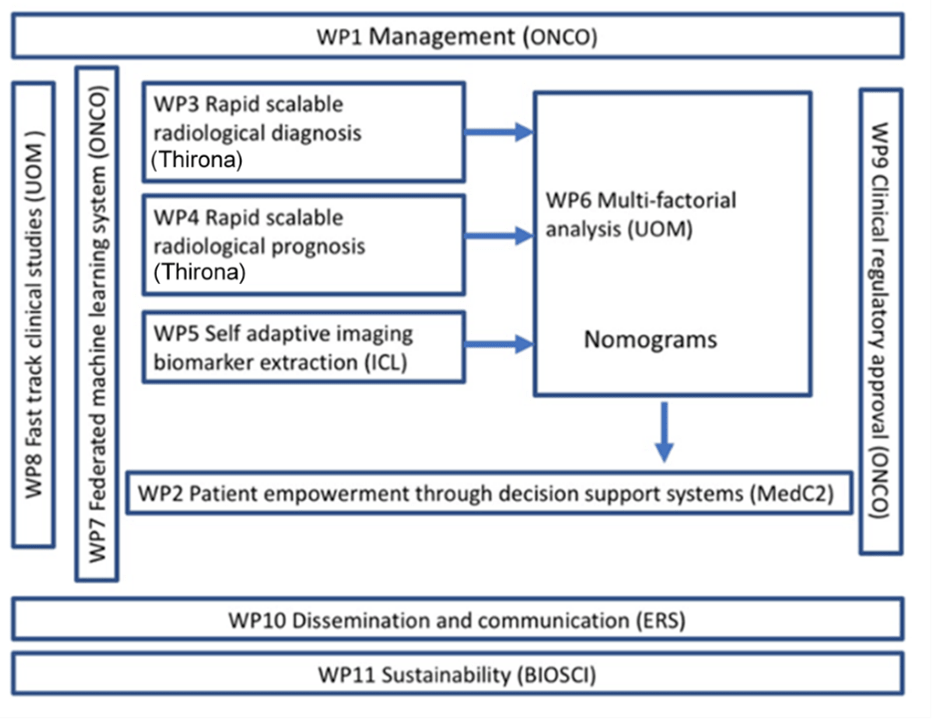
WP1: Management
The aim of this work package was to ensure the conduct of an efficient and productive consortium project by facilitating cohesiveness across the consortium through a high level of interaction. This is meant to maximise the value of public/private partnership. This work package also ensured that the governance of the project is followed and that the consortium adheres to ethics principles and manages the intellectual property produced.
WP2: Patient empowerment through decision support systems
The main objective of this work package was to enable the empowerment of patients and citizens so that they can become a societal level resource that works together with other stakeholders to help diagnose and prognosticate to optimize COVID-19 care. It was also about empowering the public to participate in data collection to improve clinical care and enable the development of precision medicine approaches that will increase the usefulness of the many new therapies under development. Patient empowerment is a process to help people gain control, including people taking the initiative, solving problems, and making decisions. As such, this work package was where the outputs of the other work packages were collected together into a multistakeholder decision support system among others aimed at patient empowerment.
WP3: Rapid scalable radiological diagnosis
This work package delviered a CE marked software suite as a medical device based upon existing solutions currently successfully deployed in Europe. They (re)trained and validated machine learning models that learn from a wide variation of clinical data, from approximately 500 coronavirus (PCR positive) patients, with varying quality and acquisition protocols. The models were trained to improve the current task of severity quantification and will be used for radiological diagnosis of COVID-19. Special attention was given to generalization of models during the development in order to successfully use the software for patients in other regions.
WP4: Rapid scalable radiological prognosis prediction
WP4 aimed to analyse the “in-hospital” longitudinal imaging data (both CXR and CT), together with other clinical parameters, to predict rapid disease progression within the next 72 hours. This is of importance in planning emergency healthcare services such as ICU space and ventilating equipment, as well as of epidemiological value such as fatality prediction. Using CT as a frontline diagnostic tool during the breakout was evaluated in this WP. Machine learning based prediction tools were developed to assist decision making and resource prioritisation. In addition, combining information extracted from sequential CXRs in their first 24-48 hours following admission with their CT scan data during model training will improve prognostic capabilities of our machine learning model further.
WP5: Privacy-preserved and self-adaptive imaging biomarker extraction
This WP investigated how to optimally adapt image quality and image information content for machine/radiomics use (as compared to visual inspection by a human). An iterative process of development, testing, assessment will be followed to deliver a validated and refined model closely linked to the aims for data harmonisation for the multicentre and multi-scanner studies of the CXR/CT images acquired for the coronavirus patients. Based on the consensus clinical protocol recommendation for image acquisition and synthetically harmonised images, image acquisition was iteratively tailored to each grading of the coronavirus patients to facilitate the most important quantitative imaging biomarkers feature extraction. To this end, image acquisition was systematically evaluated and varied to approach the optimal acquisition scenario for the dominant radiomics feature, leading to improved standardisation of the input images to the repository of the project.
WP6: Multifactorial analysis
The primary aim was to conduct a multi-factorial analysis to improve diagnosis of at-risk patients and to enable precision medicine approaches to patient care and new therapy development. Combining risk factors from multiple types of data (e.g., demographic, laboratory results, imaging) using advanced AI-enabled analysis, maximal value was extracted from available patient information. This work package was also about developing mechanistic insights into the clinical course of the disease, the conduct of immunological analyses, and molecular profiling. This type of data is essential for targeting new therapies to subpopulations mostly likely to benefit and for providing mechanistic insights if a clinical trial fails.
WP7: Technical development of federated machine learning system
Radiomics has developed DistriM (distributed machine learning) for radiology in oncology purposes. The purpose of DistriM is to continuously extract and apply updated knowledge from routine clinical care data rather than be exclusively ‘locked’ to the original clinical trial evidence. DistriM achieves this in line with the recommendations outlined by the FDA white paper on AI software as a medical device. Specifically in this project, the objective was to set up and optimise infrastructure for federated machine learning for radiology in respiratory / coronavirus. DistriM delivers individual privacy-by-design for data management / processing and is transparently auditable by a blockchain. This WP was completed successfully once the federated machine learning infrastructure was set up, optimised, validated, and clinically deployed. DistriM was populated with data from hospitals, research institutes, databases and continuously improved models are learned from these data on a regular basis. Specifically, an implementation supporting distributed learning for standardised imaging biomarker extraction, and for facilitating distributed radiomics and deep learning in medical image analysis was developed, which was utilized and integrated in all other WPs.
WP8: Fast track clinical studies
WP8 served as a rich repository of different kinds of data. This WP complemented the other work packages by investigating the diagnostic and prognostic value of biomarkers from blood, saliva, or exhaled aerosols. Data from existing hospital cohorts is combined with new cohort studies. In addition, data from the general population is collected with a user-friendly app. WP8 provided data to other WPs for (1) comparison of imaging biomarkers to other markers, (2) validation of imaging biomarkers, and (3) integration of all information in a precision medicine approach.
WP9: Accelerated regulatory approvals
This work package detailed the work necessary to rapidly develop, certify, and deploy clinically viable, fit for purpose solutions (such as a patient decision aid App or an AI model prospectively validated that can automatically detect and classify coronavirus patients in a variety of imaging settings).
WP10: Dissemination and communication
To ensure that the impact of DRAGON is maximized, WP10 exclusively focused on dissemination of results as well as communication to the public. European Respiratory Society (ERS) and European Lung Foundation (ELF) led these activities, in close collaboration with the whole consortium. A number of tools, events and policies were developed for this purpose, while a range of channels were used to reach the relevant target audience for each activity.
WP11: Sustainability
Addressing the coronavirus pandemic and preparing for future outbreaks requires societal level collaboration that delves beyond flattening the curve to increase the capacity of the entire health, research, and innovation ecosystem. Though potential exists, little has been done to improve the efficiency and predictive power of diagnostic/prognostic tests, to apply precision medicine to the patient evaluation process, or to empower patients with the ability to co-manage their disease. Building a stakeholder community around this shared vision and the project outputs will form the basis for long-term sustainability deliberations.
The focus of this work package was to develop a sustainability plan beyond the funding period and initiate its implementation. A Sustainability Working Group (SWG) was established and included representative and relevant stakeholders from within the consortium. A validated set of business models for the overall consortium and the know-how and assets were created using lean business model development methodology. Through a process of collaborative and iterative design cycles, a complete sustainability plan was developed. In addition, towards the end of the funding period, efforts were made to implement the initial steps of the sustainability plan.
WP12: Ethics
This work package sets out the ethics requirements defined by the European Commission that the project must comply with.